Neonatal Extravasation
Overview and Algorithm for Evidence-Based Treatment
NAINR. 2013;13(4):189-195.
Complete article found here: http://www.medscape.com/viewarticle/817907?nlid=45543_785&src=wnl_edit_medp_nurs&uac=123963FX&spon=24
Abstract and Introduction
Abstract
The peripheral intravenous (PIV) catheter is the most used vascular access device for the administration of medications in hospitalized neonates, however 95% of PIV catheters are removed due to complications. Infiltration and extravasation are one of the most destructive complications to the neonate's fragile skin. This article reviews multiple aspects of infiltration and extravasation injury. First, starting at the cellular level the role of vesicants in vascular injury and its role triggering inflammation will be discussed, followed by a comprehensive review of vesicants and their mechanism of injury, by pH, osmolality or chemical composition, then an overview of the NICU nurses knowledge and actions to prevent infiltration and ending with the use of an evidence-based algorithm that was developed at one children's hospital to minimize injury caused by extravasations through targeted, prompt treatment.
Introduction
Many medications given to neonates have the potential to injure when an extravasation occurs. An extravasation is described by the Infusion Nurses Society (INS) as the inadvertent administration of a vesicant solution or medication into the surrounding tissues.[1] A vesicant is defined as a solution or medication that causes the formation of blisters leading to tissue necrosis and sloughing. Extravasation can result in varying degrees of localized tissue injury and can cause pain, infection, and partial to full thickness tissue loss involving muscles and nerves. If extravasation is severe and depending on the site, skin grafts, long hospitalization and high costs result. Not surprisingly, with disfigurement and loss of function, parents may try to recover payment for their loss by initiating lawsuits.[2,3]
Infiltration Is Common Among Neonates
The peripheral intravenous (PIV) catheter is the most used vascular access device for the administration of medications in hospitalized neonates; however 95% of PIV catheters are removed due to complications such as leaking, occlusion and infiltration.[4] Infiltration rates among neonates are as high as 57%–70% with extravasation occurring in 11–23%.[5] Both infiltration and extravasation are destructive, causing localized injury to the neonate's fragile skin.[6] Infiltration of non-vesicants can cause considerable tissue damage from pressure on internal structures as additional fluid collects around the vein, and in severe cases can result in compartment syndrome.[6] Extravasations have the potential to cause peripheral tissue injury depending on the type of vesicant, concentration of the vesicant, location, amount, and duration of exposure to the vesicant. Damage from a vesicant may progress over time and become evident 48–72 hours after the extravasation occurs.[7,8]
Neonatal Vulnerability to Vascular Injury
The preterm and sick neonate is more susceptible to skin injury and complications from extravasation injury than their mature, healthy counterparts. Their immature skin structures, flexible subcutaneous tissue, small blood vessels and poor venous integrity increase the risk of complication from venipuncture and IV infusions.[5,8]The goal in neonatal care is to prevent skin breakdown whenever possible. Similarly, attention to thermoregulation, pain and stress that infants endure as a result of repeated IV attempts or restarts, and infiltrations and extravasations must be considered and managed.[9,10] Multiple tools are available to score pain responses and enable the NICU (neonatal intensive care unit) nurses to manage this appropriately. Nonpharmacologic measures to decrease pain include the use of a pacifier, swaddling, or administration of sucrose during the insertion of an IV or for infiltration and extravasation injury.[10] Aside from non-pharmacologic interventions, treating infiltration and extravasation pain with analgesics should be considered.[8]
Inflammation in the Premature Infant
The neonatal immune system is poorly regulated compared to adults and dysregulation is magnified when neonates are born early.[11-13] While intravenous therapy is necessary in this population, it is not without its risks. Vesicants can harm the endothelial lining of the blood vessel, triggering production and release of oxygen free radicals that spur inflammation.[14,15] The release of the locally induced free radicals into the circulatory system when not controlled can trigger a system-wide response to the stressor and further free radical release. In a normal response, the infant's body mounts an anti-inflammatory release of free radical scavengers. When this anti-inflammatory response is inadequate, which is common in prematurity, or the inflammatory assault is severe, endothelial dysfunction leads to programmed cell death (apoptosis).[16] The load of oxidative stress in premature infants is especially of concern as it has been linked to various neonatal morbidities including necrotizing enterocolitis,[16,17] retinopathy of prematurity,[18-20] and chronic lung disease.[16,21-23]
Pathophysiology of Vascular Injury
In the human body, vascular injury, oxidative stress, and inflammation are intricately related. Infiltration and extravasation are both symptoms of vascular injury. When vascular injury occurs, the release of free radicals is stimulated and the energy producing ability of the cell becomes dysfunctional as mitochondria are damaged. As free radicals are released and the capacity of the damaged mitochondria to produce energy is limited, the balance between nitric oxide and superoxide is disrupted. This reactive oxygen species excess extends the damage.[24-26] A continual cycle of free radical production and endothelial apoptosis occurs leading to injury of cell membranes and vessels. Symptoms of this process include redness, swelling (inflammation with increasing vascular permeability), and visible tissue injury.[25] The inflammatory process involved in extravasation is depicted in Fig 1. Note how the interaction of the biological response to chemical and non-chemical risk factors when exposed to the harmful drug directly affects healthy endothelium, leading to vascular injury. The framework is a helpful tool in understanding the biological aspects of extravasation and action targets to prevent injury.
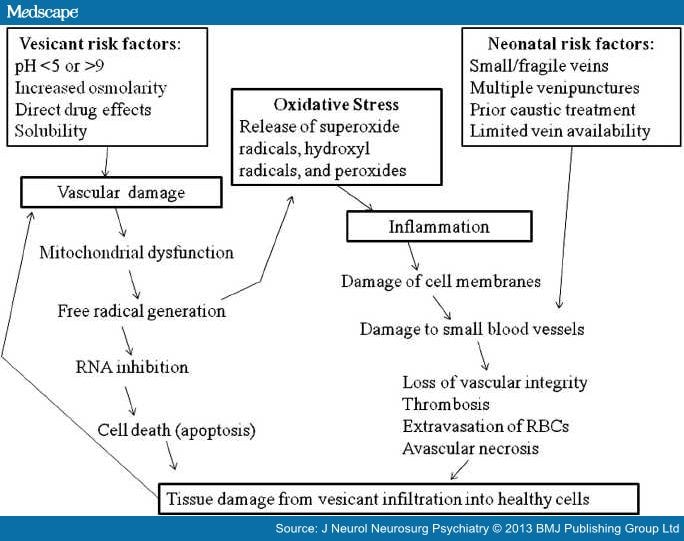
Figure 1.
Model of vascular injury.
The Neonatal Intensive Care Unit (NICU) Nurse's Role
NICU nurses monitor the PIV site with vigilance to aid in early identification of infiltration and extravasation and prevent this type of injury whenever possible. Identifying an infiltration may be difficult, even for the most experienced nurse.[10] The NICU nurse is aware of the subtle changes in heart rate, oxygen saturations, apnea, and the more obvious change in behavior such crying and agitation that may indicate problems with the PIV therapy.[4] Neonatal nursing entails not only basic knowledge of the anatomy and physiology of neonatal skin and how to prevent iatrogenic injury from routine nursing care but also knowledge of vascular anatomy and infection control for the safe delivery of infusion therapy.[4,8,10] Several organizations have developed standards and guidelines that assist nurses with the best practice in infusion therapy.[10] These evidence-based guidelines and grading scales for infiltrations can aid with standardizing documentation and protocols that guide nursing care and improve patient outcomes.[5,6]
Potential Origins of Infiltration
There is a supposition that an infiltration or extravasation is caused by IV catheter dislodgement or puncture of the vein during insertion or during handling of the infant. Chemical composition of medications also impacts risk of vein rupture.[5] The vein's tolerance to an infusion is affected by the osmolality and pH of the vesicant, the duration of the exposure, and irritation to the endothelial cells.[4] An additional factor in causing a cannulated vessel to rupture and leak is the pressure in which the medication is being delivered by the infusion pump.[3,5] Janet Pettit describes the following 3 theories of the mechanism of infiltration and extravasations. The first theory states that the irritation of the venous endothelium from the infusate causes vasoconstriction and diminished blood flow.[4] The second theory has been confirmed by dye studies. It shows infiltration of the infusate through the catheter insertion hole created with IV placement; this can occur when the flow proximal to the catheter tip becomes obstructed.[4] The third theory is that irritation of the venous endothelium caused by the osmolality, pH, or chemical composition of the infusate damages the lining of the vein and allows transmission of the infusate into the tissue, without creating a puncture in the vein.[4] These three theories demonstrate how infiltration and extravasation can occur; the remainder of the damage is thought to be caused by the irritants and vesicants.
Irritants and Vesicants Given to Neonates
Intravenous medications can be divided into three major subcategories: 1) non-vesicants, 2) irritants, and 3) vesicants. In order for an infiltration to be a true extravasation, the offending agent, by definition, must be a vesicant. There are a number of different qualities that affect the potential for a medication to result in tissue damage. These include, but are not limited to: osmolarity, pH, direct medication effects and solubility.[27] See for medications categorized by their mechanism of injury.
Table 1. Medication Mechanisms of Vascular Injury.
| Inciting Agent | |||||||
|---|---|---|---|---|---|---|---|
| Vesicants (phenylephrine) | Irritants | Hypo-osmolar | Hyperosmolar (>10% dextrose, mannitol, potassium, propofol and sodium bicarbonate) | Alkalotic Medication | Vasoactive Medication | Highly-Lipophilic Medications | |
| Definition | Medications capable of causing tissue damage9e.g. Calcium stimulates smooth muscle to contract capillaries, leading to hypo-perfusion and ischemic injury | Causes pain within the vessel or surrounding tissue but does not result in tissue necrosis | Osmolarity < 280 mOsmcausing cells to swell as fluid shifts into the cell. As the intracellular volume exceeds its capacity, cells can lyse. | Osmolarity > 280 mOsm, causing cells to shrink as fluid shifts outside of the cell | High concentration of hydrogen ions (high pH) causes inflammation and can lead to vascular injury | Alpha-receptor stimulation constricts capillary beds, decreases local blood flow and deprives local tissue of oxygen leading to ischemic injury5 | Medications do not dilute well in water, making it difficult to flush out or wash off, damage results from the high concentration of the medication in the tissue |
| Medication Examples | Acyclovir Aminophylline Calcium Chloride Dobutamine Dopamine Epinephrine Nafcillin Norepinephrine Oxacillin Penicillin Phenytoin, Potassium Salts Total parenteral nutrition (TPN) Peripheral parenteral nutrition (PPN)Vancomycin3 | Aminophylline Calcium Gluconate Digoxin, Erythromycin, Gentamicin, Theophylline3 | 0.2 % NaCl Sterile water5 | 3% sodium chlorideCalcium chlorideContrast mediaTotal parenteral nutrition5 | Sodium Bicarbonate Phenobarbital Sodium thiopental Phenytoin | Dobutamine Epinephrine Norepinephrine Vasopressin | Diazepam Digoxin Nitroglycerine Phenytoin |
Osmolarity describes the number of particles which are suspended in solution. Normal serum osmolarity of a neonate is approximately 280 mOsm. When a solution has a higher osmolarity, it is considered to be hyperosmolar. The effect causes cells to shrink as fluid shifts from inside to outside of the cell. This response occurs in order to increase the osmolarity inside the cell to equilibrate with the elevated osmolarity of the serum. The opposite of this effect would be a solution that has a lower osmolarity or is hypoosmolar. In this situation, fluid would enter the cell in order to decrease the osmolarity of the cell to equal that outside. At the most extreme, this can result in cell lysis as the volume of the cell exceeds its capacity. Osmolarity may be altered by how a medication is diluted. Ampicillin is a good example of this. If 50 mg of ampicillin is mixed with 1 mL of sterile water, the osmolarity is 243. If 50 mg of ampicillin is mixed with 1 mL of normal saline, the osmolarity would be increased to 493–520 and thus increase potential damage to tissue.[4] It is generally felt that solutions with an osmolarity < 900 mOsm can be administered peripherally. This is based on an American Academy of Pediatrics Committee on Nutrition recommendation governing the use of peripheral parenteral nutrition (PPN).[4] There is some controversy with this recommendation due to a lack of supporting evidence. The Infusion Nurses Society recommends limiting the osmolarity of peripherally administered solutions to < 500 mOsm. This is based on evidence that more concentrated solutions are more likely to result in chemical phlebitis.[4]
The measurement of the concentration of hydrogen ions, pH, is another important consideration when evaluating a medication's risk of infiltration or extravasation. A normal pH of blood is between 7.35 and 7.45 (arterially). As the pH of a medication moves away from this normal range, the risk of tissue injury is higher. As the number of hydrogen ions increase, the solution will become more acidic (pH < 7.35). The majority of medications have an acidic to neutral pH. Once medications move out of the range of 5–9, risk of inflammation and vascular injury increases significantly. Vancomycin, for example, has a pH of 2.5–4.5, which helps to explain its potential for extravasation. Medications with alkalotic pH (> 7.45) can be just as damaging to tissue. One example is phenytoin with a pH of 10–12, approximating that of household bleach.
Medications have specific mechanisms of action that may result in cellular damage when an infiltration/extravasation occurs. Vasoactive medications such as dopamine have effects on alpha-receptors. If this product is introduced into tissue, this alpha stimulation will result in constriction of capillary beds, which subsequently will decrease local blood flow. Local tissue will be deprived of oxygen and ischemic injury will ensue.[4] Another group of medications that may have direct effects on tissues are electrolytes. Calcium is necessary for depolarization and contraction of smooth muscle. If a concentrated calcium solution is infused into tissue, this may result in capillary constriction via stimulation of smooth muscle, which will also result in ischemic injury to hypoperfused tissue.[4,7] Finally the solubility of a medication can influence its contribution to local tissue damage. The more lipophilic a medication, the less water-soluble it is. This makes washing out the site impractical and leads to high concentrations of medications located in relatively small areas of tissue that can lead to concentration dependent direct tissue damage.
Nursing Actions to Prevent Vascular Injury
The best method to decrease complications of PIV therapy is to prevent them in the first place.[2] Serious complications are not entirely preventable, but following recommended standards of IV therapy is the best approach for avoiding complications.[3] The decision to place a peripherally inserted central catheters (PICC) or central venous lines (CVL) might be needed if vascular access is difficult or long-term parenteral therapy is planned. However, this may be difficult in low birth weight (LBW) neonates when access for a CVL is limited, central line infection prevention initiatives are paramount, or a variety of incompatible infusions are indicated.[2,6] The risks and benefits of PICC and CVL are evaluated by the medical and surgical teams. If the risks are reasoned to be too great for PICC or CVL, responsibility will be on the nurse to manage the PIV and when vesicants are being infused vigilance needs to heighten. Evidence-based guidelines for infusion therapy start with the insertion. See for recommendations to prevent peripheral vascular injury. On insertion, attention needs to be paid to the selection of the appropriate catheter, site selection, skin antisepsis and stabilization once the device is in place.[10] NICU nurses learn to leave the neonates' extremity out of the swaddling or sleep sacks for easy visualization of the PIV site; so that hourly assessments (and more frequent assessments if a known vesicant is infusing) do not interrupt the neonate's developmental sleep. Warning signs of possible complication warranting further investigation include a PIV that is difficult to flush or an infant who responds to the flush with crying or flinching.[3] Assessments might reveal swelling, redness, and coolness of the skin. Most infiltrates resolve spontaneously after the IV catheter is removed, however in severe cases blistering and skin necrosis, prolonged capillary refill and decreased pulses and movement of the extremities can result. Infiltration scales can be useful to nurses in identifying the severity of the assessment findings.[3]
Table 2. Recommendations for Practice to Prevent Vascular Injury.
| Peripheral IV Insertion and Maintenance |
| Use small enough plastic/silicone catheter to avoid restriction of blood flow |
| Avoid repeated use of a vein |
| Avoid placing a PIV in an areas difficult to immobilize |
| Use transparent tape to secure |
| Cover the site with a sterile semi-permeable transparent dressing that will permit ongoing visualization of the insertion site |
| Upper extremities less likely to infiltrate or leak compared with peripheral IV in lower extremities or scalp veins |
| Place tape loosely over boney prominences to avoid restricting blood flow to the extremity |
| Infusion Maintenance |
| Limit PIV glucose to 12.5% |
| Dilute medications as much as possible before administration are other solutions to preventing extravasation |
PIV= peripheral intravenous.
Key Actions to Minimize Injury When Extravasation Occurs
Once an infiltration or extravasation is discovered, immediate treatment is the key to preventing progressive damage from the vesicant.[2] Treatment decisions are based on the size and appearance of the injury, type of IV infusing, duration of exposure and location.[8] Protocols and algorithms can be used to assist nurses in the steps needed to minimize the potential damage and start the treatment process. Stopping the infusion and elevation of the extremity is the first actions followed by placement of a saline soaked gauze or prepackaged normal saline pad. The saline draws out the vesicant, and impedes a scab from forming to allow fluid to leak out. Gently squeezing the fluid from the open insertion site can also help to remove the offensive agent.[8] While the saline soaks are held in place, assistance with the various tasks that are required for treatment may require additional personnel. A new PIV may need to be inserted immediately, the physician or nurse practitioner (NP) will be called to assess the site and plan for pharmacologic treatment if indicated. For an extravasation, treatment should be determined prior to catheter removal.[8]The administration of antidotes or enzymes such as hyaluronidase is the method of choice to manage IV extravasation.[6] Treatment that improves tissue perfusion and prevents progressive tissue necrosis has been shown to be most effective if initiated within 1 hour.[8] Ongoing assessment of the neonate and measures to improve comfort or alleviate pain may be indicated. Compassionately reporting the situation to the family as soon as possible and keeping them up to date on the plan of action are required.[8] Prior exposure to standards of care, protocols and algorithms and staff education could aid the process of administering antidotes, as this could be time consuming "on-the-job-training" for an inexperienced NICU nurse, when timely treatment provides better patient outcomes.[8] An algorithm based on the work of Sawatzky-Dickson and colleagues (2006) (see Fig 2) was developed to streamline the described process and improved patient outcomes.[2]
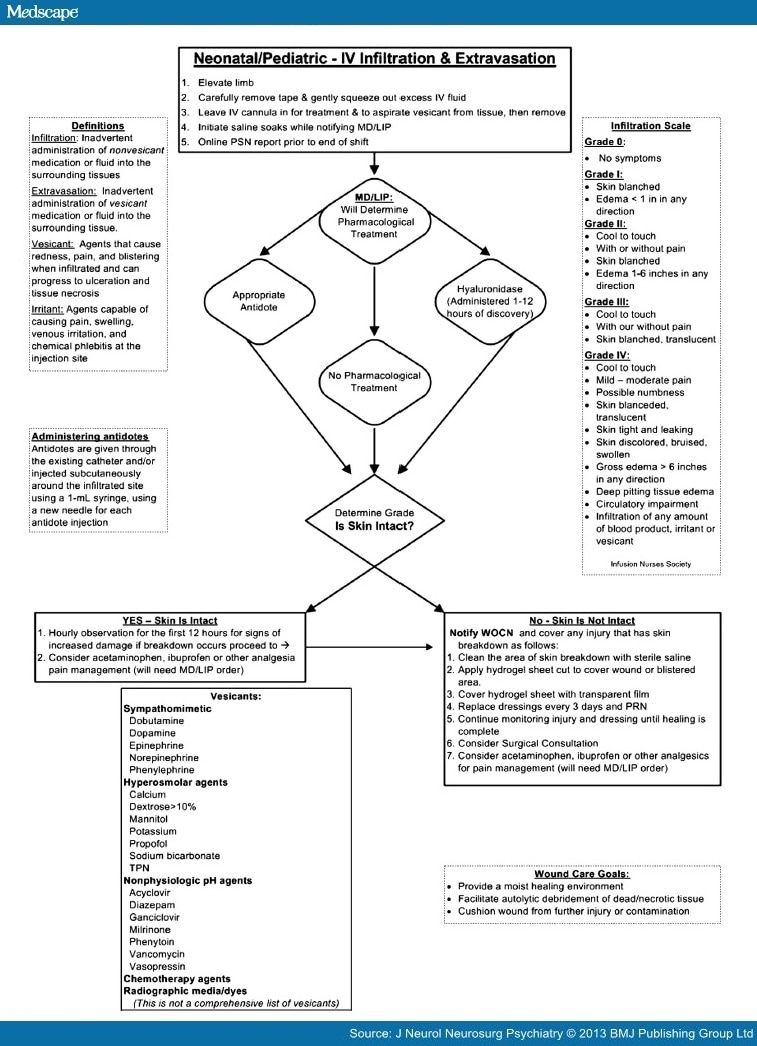
Figure 2.
Diamond Children's IV Infiltration and Extravasation Algorithm (adapted from Sawatzky-Dickson and colleagues, [2006]). LIP = Licensed Independent Practitioner, MD = Medical Doctor, WOCN = Wound, Ostomy, and Continence Nurse.
The quality improvement initiatives implementing the algorithm have not been formally evaluated. However, physicians, pharmacists and nurses are managing infiltration and extravasations promptly as a team. The administration of hyaluronidase or the appropriate antidote without the WOCN's assistance, as well as a decline in referrals for long-term wound management for extravasation injuries are now the NICU's norm.
Pharmacologic Options to Treat Extravasations
Currently there are multiple medications that are used to treat extravasations. These include hyaluronidase, phentolamine, and topical nitroglycerin 2% ointment.[8]Sodium thiosulfate, dimethyl sulfoxide (DMSO) and dexrazoxane are antidotes primarily associated with treatment of chemotherapy extravasations. As the scope of this article is directed at the neonatal population, it will focus on the first three non-chemotherapeutic extravasation medications: hyaluronidase, phentolamine and nitroglycerin.[7,8,26,27]
Hyaluronidase
Hyaluronidase is an enzyme that breaks down hyaluronic acid, a compound best described as the "glue" which holds cells together. When this extracellular glue is dissolved, cells are separated. This is helpful in extravasations as it allows for the medication to distribute through a larger area by breaking down the walls that keep it localized. This not only helps to decrease the concentration effects of the extravasated product, but also exposes the medication to more capillary beds that allows for reduction of edema via more rapid reabsorption and removal of the product from the damaged area. The overall effect is to minimize the local damage caused by the extravasation. This change is not permanent, as hyaluronic acid will be regenerated within a 24 to 48 hour period. Efficacy of hyaluronidase is supported by an FDA subcommittee evaluation from 2009 based on evidence from animal studies, human studies and clinical case reports. Hyaluronidase has been used to limit extravasation injury since 1976. One study suggested that use of hyaluronidase may decrease the development of skin ulcers by 50–60% and decrease skin ulcer size by as much as 50%.[7,28,29]
Hyaluronidase has been used in various ways and consensus is lacking on which way is best. Most protocols advocate administration of hyaluronidase within 1 to 2 hours of the initial injury but there are case reports suggesting administration as far out as 10 days may be beneficial. Concentrations of hyaluronidase used in studies ranges from 15 unit/mL to 1500 unit/mL but standard practice for neonatal and pediatric patients is to use a concentration of 15 unit/mL. Some centers will have the practitioner pull back the infiltrated IV 1 to 2 mm to avoid IV injection and then administer 0.2 mL of a 15 unit/mL solution through the catheter to deliver hyaluronidase directly to the site of extravasation. The standard way to utilize hyaluronidase is to draw up 5 syringes of 0.2 mL of the 15 unit/mL concentrations and, after cleansing the site, inject these at 5 separate sites around the area of extravasation. Again, the idea is to break down the barrier that is holding the medication in that specific area and allow it to disperse to a larger surface area.[26,28] Adverse effects are uncommon but potentially include tachycardia, hypotension, dizziness, chills, urticarial erythema, angioedema, nausea and vomiting.
Phentolamine
Phentolamine is an antidote that will counteract the effect of vasoactive agents such as dopamine, epinephrine, norepinephrine and phenylephrine.[8] These medications result in vasoconstriction via stimulation of alpha-receptors. Phentolamine acts to block the activity of alpha-receptors and subsequently will help relax vascular smooth muscle. This will improve circulation in the area of the extravasation and thus decrease ischemia and cell death. Phentolamine can also be utilized for vasopressin or dopamine extravasation.[28] Phentolamine should be administered within 12 hours of initial exposure but administration should occur as soon as possible. Prepare a 0.5 to 1 mg/mL solution and administer 0.1 mg/kg (to a max of 2.5 mg in neonates, 10 mg in older children and adults) into the existing IV of the affected area. Neonates should be monitored closely for potential development of hypotension, dysrhythmias and tachycardia due to potential systemic absorption of phentolamine.[3,28] The efficacy of phentolamine has been demonstrated in animal studies and case reports.[30,31,32]
Nitroglycerin Ointment (2%)
Nitroglycerin 2% is an option to treat extravasations.[8] Nitroglycerin acts to relax smooth muscle resulting in arteriolar, arterial and venous vasodilation that results in increased capillary blood flow, counteracting the effects of vasoactive medications. This will help to reverse tissue ischemia and cell death. In the neonatal population, there is a case report describing the use of 1 inch of 2% nitroglycerin ointment for treatment of a dopamine extravasation, located in the dorsum of the left hand, in a 1.8 kg 34 week preemie. This resulted in return of circulation within a few minutes. Of note, treatment was started almost 12 hours after the extravasation was initially noted and patient had no significant change in hemodynamics.[33]
Ongoing Assessment and Wound Care
If the skin is intact, elevation of the extremity and hourly observation for the next 12 crucial hours will aid in resolution of the edema and early identification for the development of surface skin injury. If there is blistering or skin breakdown; moist wound healing is indicated. The days that these injuries were left open to dry out and heal on their own should be gone and moist wound healing methods took their place. A moist wound environment will facilitate epidermal regeneration, fibroblast proliferation, and endothelial cell proliferation.[2] Now with advanced wound care treatments; healing can be safe and atraumatic for the neonate with minor skin breakdown or in the neonate with full thickness wounds requiring wound care for weeks. Hydrogel dressings are one of the moist wound healing treatments that have been found to be evidence-based and safe for neonates of all gestations.[8,9] Hydrogels consist of 80–90% water, which can be soothing and gentle to skin and keeps the wound moist to facilitate auto-debridement of wounds by rehydrating sloughing tissue and enhancing the rate of autolysis.[8] A section of hydrogel sheet cut to cover the wound and secured with transparent dressing reduces dressing changes to every 3 days, resulting in decreased handling and discomfort of the neonate, and less trauma to regenerated tissue, as well as protects the tissue from outside oxygen tension and provides a lower pH, which inhibits the growth of pathogens.[2,3] Full thickness injury may require surgical debridement and skin grafting, by a pediatric or plastic surgeon.[8] Adhesive silicone foam can be used for shallow wounds that may only require a small daily application of amorphous gel to maintain moisture to the site. The moisture of hydrogels at times can macerate the periwound, in infants greater than 30 days; an alcohol free skin barrier can be applied to protect the fragile skin surrounding the wound.[8]
Conclusions
Preventing and detecting early damage from vesicants are a priority of every neonatal nurse. In this article we have described the pathophysiology of vascular injury, the medications that incite it and recommendations to prevent and treat infiltrations and extravasations. Incorporating training on critical nursing assessments, signs of injury and key actions to treat it are an important part of every NICU's education plan. We recommend using an algorithm, like the one presented here, to guide actions once extravasation occurs. Using non-pharmacologic approaches to treat the injury is a first step in the process of limiting damage. For certain vesicant-induced injury, pharmacologic treatment using hyaluronidase, in particular, has been shown to limit the injury. If ineffective or the injury is extensive, surgical treatment may be necessary and a plastic surgeon may need to be consulted. Along the way, openly speaking with the parents about the treatment is necessary and important to maintain the family-healthcare partnership.